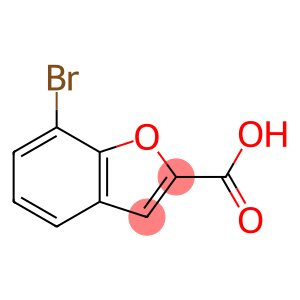
2-benzofurancarboxylic acid, 7-bromo-
7-Bromo-1-benzofuran-2-carboxylic acid
CAS: 550998-59-9
Molecular Formula: C9H5BrO3
2-benzofurancarboxylic acid, 7-bromo- - Names and Identifiers
| Name | 7-Bromo-1-benzofuran-2-carboxylic acid |
| Synonyms | 7-Bromo-2-benzofurancarboxylic acid 2-Benzofurancarboxylic acid, 7-bromo- 2-benzofurancarboxylic acid, 7-bromo- 7-BROMO-1-BENZOFURAN-2-CARBOXYLICACID 7-Bromo-1-benzofuran-2-carboxylic acid |
| CAS | 550998-59-9 |
| InChI | InChI=1/C9H5BrO3/c10-6-3-1-2-5-4-7(9(11)12)13-8(5)6/h1-4H,(H,11,12) |
2-benzofurancarboxylic acid, 7-bromo- - Physico-chemical Properties
| Molecular Formula | C9H5BrO3 |
| Molar Mass | 241.04 |
| Density | 1.784±0.06 g/cm3(Predicted) |
| Boling Point | 370.9±22.0 °C(Predicted) |
| Flash Point | 178.1°C |
| Vapor Presure | 3.71E-06mmHg at 25°C |
| pKa | 2.90±0.30(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.679 |
2-benzofurancarboxylic acid, 7-bromo- - Reference Information
| Preparation | Method 1: The preparation of 7-bromobenzofuran-2-formic acid is divided into the following two steps: 1) 3-Bromo-2-Hydroxybenzaldehyde. 20.0g(115.6mmol) of 2-bromophenol was added to 500ml of anhydrous acetonitrile. 16.84g(176.87mmol) anhydrous magnesium chloride, 23.4g paraformaldehyde particles and 41.9ml(300.6mmol) triethylamine were added. The reaction mixture was heated and refluxed for 4 hours, and after cooling to 0°C, 300 ml2N hydrochloric acid was added. The aqueous phase was extracted three times each time with 200ml diethyl ether. The organic phase was dried with magnesium sulfate and the solvent was removed in vacuum. The title compounds were isolated and reacted further without further purification. 24g (64% of theoretical value, 62% purity according to HPLC). HPLC (Method 1):Rt = 4.25 min. MS(ESIpos):m/z = 202(M H). 2) 7-Bromobenzofuran-2-formic acid. 13.5g(40.3mmol) 3-bromo-2-hydroxybenzaldehyde (Example 28A,62% purity) was heated with 9.18g(84.62mmol) of methyl chloroacetate, 1.49g(4.03mmol) of tetra-n-butylammonium iodide and 22.28.G (161.18mmol) potassium carbonate was kept at 130 ℃ for 6 hours. After cooling to room temperature, add 100ml of water and 100ml of THF, and 13.57g(241.77mmol) of potassium hydroxide, and stir the mixture overnight at room temperature. Remove the solvent under reduced pressure, dissolve the residue in 400ml of water, and wash four times with a total of 400ml diethyl ether. At the same time, the pH was adjusted to 0 with concentrated hydrochloric acid and extracted 5 times with a total of 700ml of ethyl acetate. The organic phase was washed with 100ml saturated sodium chloride solution and dried with magnesium sulfate. The crude product was completely removed from the residual solvent under high vacuum and stirred with 80ml of diethyl ether. The product 7-bromobenzofuran-2-formic acid was filtered out and washed with a small amount of ice-cold ether. Output: 4.8 grams (47% of theoretical value). 1H-NMR(200MHz,DMSO-d6):δ = 13.5(br.s,1H),7.86-7.72(m,2H),7.79(s,1H),7.31(t,1H). MS(DCI/NH3):m/z = 258(M NH4). |
| application | 7-bromobenzofuran-2-formic acid can be used as an intermediate in pharmaceutical synthesis. |
Last Update:2024-04-09 02:00:09
Supplier List
Spot supply
Product Name: 7-Bromobenzofuran-2-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 550998-59-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-Bromo-1-benzofuran-2-carboxylic acid Request for quotation
CAS: 550998-59-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 550998-59-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 7-BROMO-1-BENZOFURAN-2-CARBOXYLICACID Request for quotation
CAS: 550998-59-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 550998-59-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 7-BROMO-1-BENZOFURAN-2-CARBOXYLICACID Visit Supplier Webpage Request for quotation
CAS: 550998-59-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 550998-59-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 7-Bromobenzofuran-2-carboxylic acid Visit Supplier Webpage Request for quotationCAS: 550998-59-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-Bromo-1-benzofuran-2-carboxylic acid Request for quotation
CAS: 550998-59-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 550998-59-9
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 7-BROMO-1-BENZOFURAN-2-CARBOXYLICACID Request for quotation
CAS: 550998-59-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 550998-59-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 7-BROMO-1-BENZOFURAN-2-CARBOXYLICACID Visit Supplier Webpage Request for quotation
CAS: 550998-59-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 550998-59-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



